Aeshilo H + L Dual HA Skin Booster (1×2.5ml)
AESHILO®
Injectable hyaluronic acid skin booster for aesthetic useDual molecular weight (high and low molecular weight) non-crosslinked hyaluronic acid bio-remodelling skin booster
Certifications
- Manufactured in facilities operating under Good Manufacturing Practice (GMP) with permissions from the Korean FDA, as stated by the producer and trading partner
- Production and quality management reported as compliant with ISO 9001, ISO 13485 and ISO 22716 standards
- Described as having cosmetic certification from Korean authorities (KFDA Equivalent cosmetic grade) for export as a cosmetic/skin booster product
- Stated to have European CPNP cosmetic registration, indicating compliance with EU cosmetic Product notification requirements
- Classified and traded internationally under HS Code 3304991000 (beauty or make Up preparations and preparations for the care of the skin), consistent with cosmetic/skin booster categorisation rather than licensed medicinal product or CE Marked medical device
- Manufactured in facilities operating under Good Manufacturing Practice (GMP) with permissions from the Korean FDA, as stated by the producer and trading partner
- Production and quality management reported as compliant with ISO 9001, ISO 13485 and ISO 22716 standards
- Described as having cosmetic certification from Korean authorities (KFDA Equivalent cosmetic grade) for export as a cosmetic/skin booster product
- Stated to have European CPNP cosmetic registration, indicating compliance with EU cosmetic Product notification requirements
- Classified and traded internationally under HS Code 3304991000 (beauty or make Up preparations and preparations for the care of the skin), consistent with cosmetic/skin booster categorisation rather than licensed medicinal product or CE Marked medical device
Dual molecular weight (high and low molecular weight) non-crosslinked hyaluronic acid bio-remodelling skin booster
Description
Aeshilo H + L Dual HA Skin Booster (1×2.5ml) is an injectable skin booster based on a hybrid composition of high molecular weight (H-HA) and low molecular weight (L-HA) hyaluronic acid of non-animal origin. It is designed as a bio-remodelling skin treatment that uses a thermal hybrid complex HA technology (HILO Hybrid composition Technology) to improve overall skin quality rather than simply adding volume like traditional dermal fillers. According to the manufacturer and international distributors, AESHILO is formulated with highly purified medical-grade hyaluronic acid in a non-crosslinked hybrid form to enhance skin hydration, firmness, elasticity and texture, helping to reduce the appearance of wrinkles and fine lines. It is intended for professional aesthetic use as an injectable skin solution, integrating easily into existing aesthetic treatment protocols with short recovery time and a safety profile supported by GMP manufacturing and cosmetic regulatory registrations.
Bnefits
- Hybrid composition of high molecular weight and low molecular weight hyaluronic acid designed for skin bio-remodelling rather than simple volumisation
- Enhances skin hydration, tone and elasticity through highly purified, non-animal origin hyaluronic acid
- Aims to improve overall skin quality, including firmness and texture, and to reduce the appearance of wrinkles and fine lines
- Uses proprietary HILO Hybrid composition Technology and thermal hybrid processing instead of chemical cross-linking agents such as BDDE
- Injectable skin solution manufactured from ultra-pure medical-grade hyaluronic acid under GMP conditions
- Marketed as having a favourable safety profile and short recovery period, making it suitable for integration into aesthetic treatment plans
- Registered as a cosmetic/skin booster product with Korean and European cosmetic frameworks (including KFDA-equivalent cosmetic grade and CPNP registration), supporting international distribution in professional aesthetic settings
Indications
- Professional aesthetic injectable treatment aimed at improving skin hydration, firmness, elasticity and texture in adults
- Bio-remodelling of multi-layer skin tissue to address signs of skin ageing such as fine lines and wrinkles
- Skin quality improvement on areas commonly treated with skin boosters (e.g. facial and potentially other aesthetic treatment areas) as per professional protocols
- Use as part of non-surgical skin rejuvenation programmes performed by qualified practitioners
Composition
- Total hyaluronic acid content reported by distributors as 80 mg per 2.5 mL (40 mg/mL high molecular weight hyaluronic acid + 40 mg/mL low molecular weight hyaluronic acid)
- Hyaluronic acid of non-animal origin, described as ultra-pure medical-grade HA
- Hybrid composition of high molecular weight and low molecular weight hyaluronic acid created via thermal hybrid processing (HILO Hybrid composition Technology)
- Non-crosslinked hyaluronic acid formulation (manufacturer highlights the absence of chemical cross-linking agents such as BDDE, relying instead on hybrid HA composition technology)
- Other excipients: Not publicly listed
Formulation
- Sterile injectable skin solution based on a hybrid complex of high and low molecular weight hyaluronic acid
- Non-crosslinked hyaluronic acid gel using thermal hybrid composition technology rather than chemical cross-linking
- Transparent or translucent viscoelastic HA formulation supplied ready for injection in a prefilled syringe
- Total volume per syringe: 2.5 mL
Packaging
- Supplied as a single prefilled 2.5 mL sterile syringe per box (packing unit stated as 2.5 mL / 1 syringe)
- Packaged for professional use in aesthetic clinics and medical settings; detailed box artwork and included accessories (e.g. needles) are not publicly specified
Usage
- Intended strictly for use by trained and appropriately qualified healthcare or aesthetic professionals experienced in injectable treatments
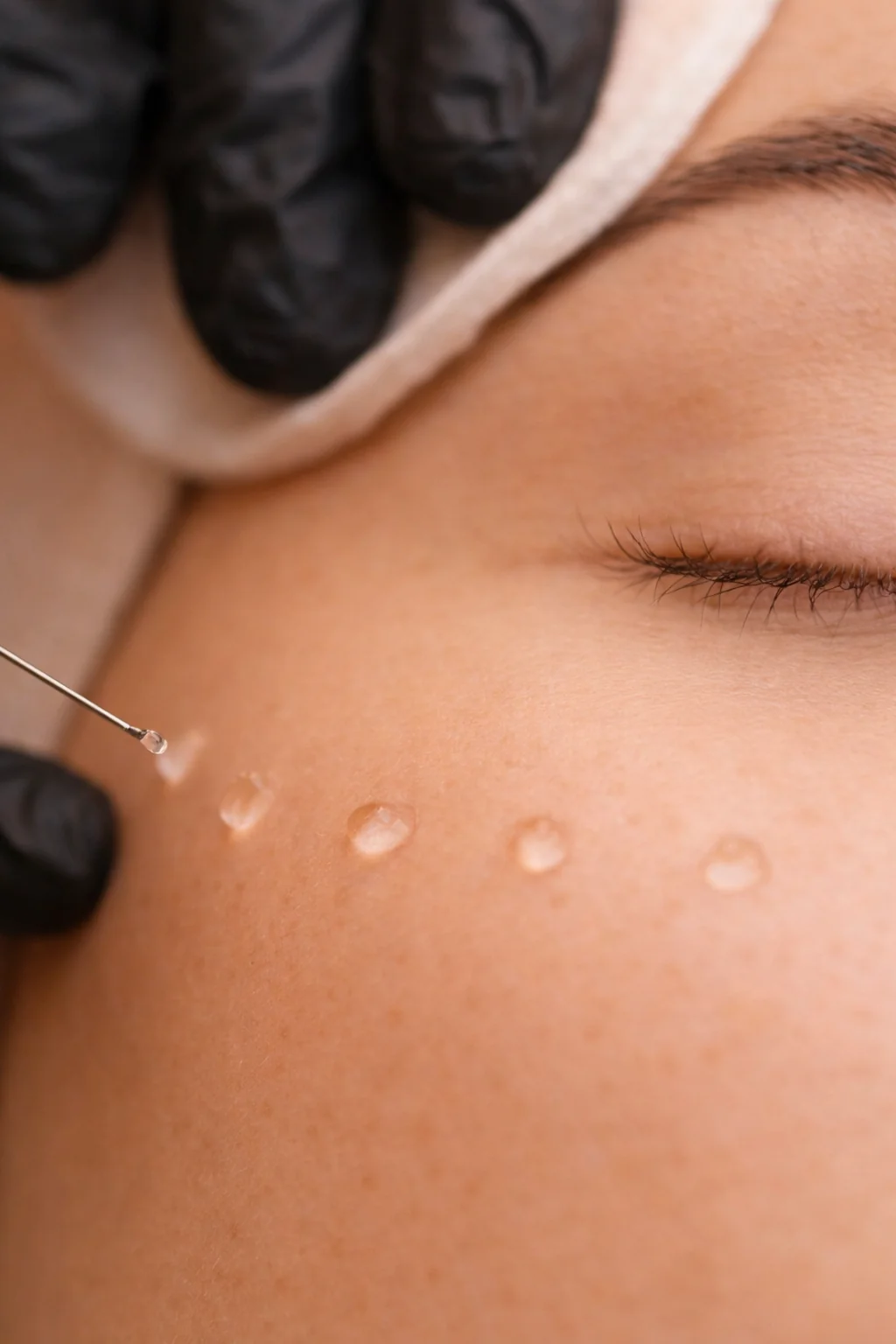
- Used as an injectable skin booster for bio-remodelling of skin tissue; specific injection techniques, anatomical sites, depth, dosing and treatment intervals must follow the official instructions for use provided with the product and local clinical guidelines
- Not for self-administration by patients or use by untrained persons
- Practitioners should follow standard aseptic technique and infection-control measures when preparing and administering injections
- Patients should be counselled regarding expected outcomes, possible risks, and post-treatment care according to the manufacturer’s directions and clinic protocols
Contraindications
- Not publicly listed (no official, product-specific list of contraindications is available in publicly accessible technical or regulatory documents; practitioners should refer to the product’s instructions for use and apply general contraindication principles for injectable hyaluronic acid skin boosters).
Adverse Effects
- Not publicly listed (no detailed, product-specific adverse reaction profile is publicly available; general risks of injectable hyaluronic acid procedures such as injection site reactions, swelling, bruising, pain, or other complications must be assessed and discussed by the treating professional using the product’s instructions for use and current clinical guidance).
Storage Conditions
- Not publicly listed (publicly accessible materials do not specify exact storage temperature range or conditions; storage instructions must be taken from the official packaging and technical data sheet supplied with the product).
Duration
Not publicly listed (no specific, standardised duration of clinical effect or recommended treatment course is stated in publicly accessible manufacturer or distributor materials; treatment schedules and maintenance intervals are determined by the treating practitioner based on individual patient assessment).
Onset
Not publicly listed (public-facing materials describe aesthetic skin quality improvements but do not provide a specific, quantified onset-of-action timeframe).