CQC compliance requires extensive documentation. From policies and procedures to risk assessments and consent forms, understanding what you need and how to maintain it properly is essential for CQC registration.
Why CQC Compliance Documentation Matters
CQC compliance documentation proves you meet CQC Fundamental Standards and provide safe care. Without proper documentation, you cannot demonstrate compliance. During CQC inspections, inspectors request specific documents—if you can’t produce them quickly, it signals poor governance.
What inspectors check: Documents are current, clinic-specific (not generic templates), actually implemented, and regularly reviewed.
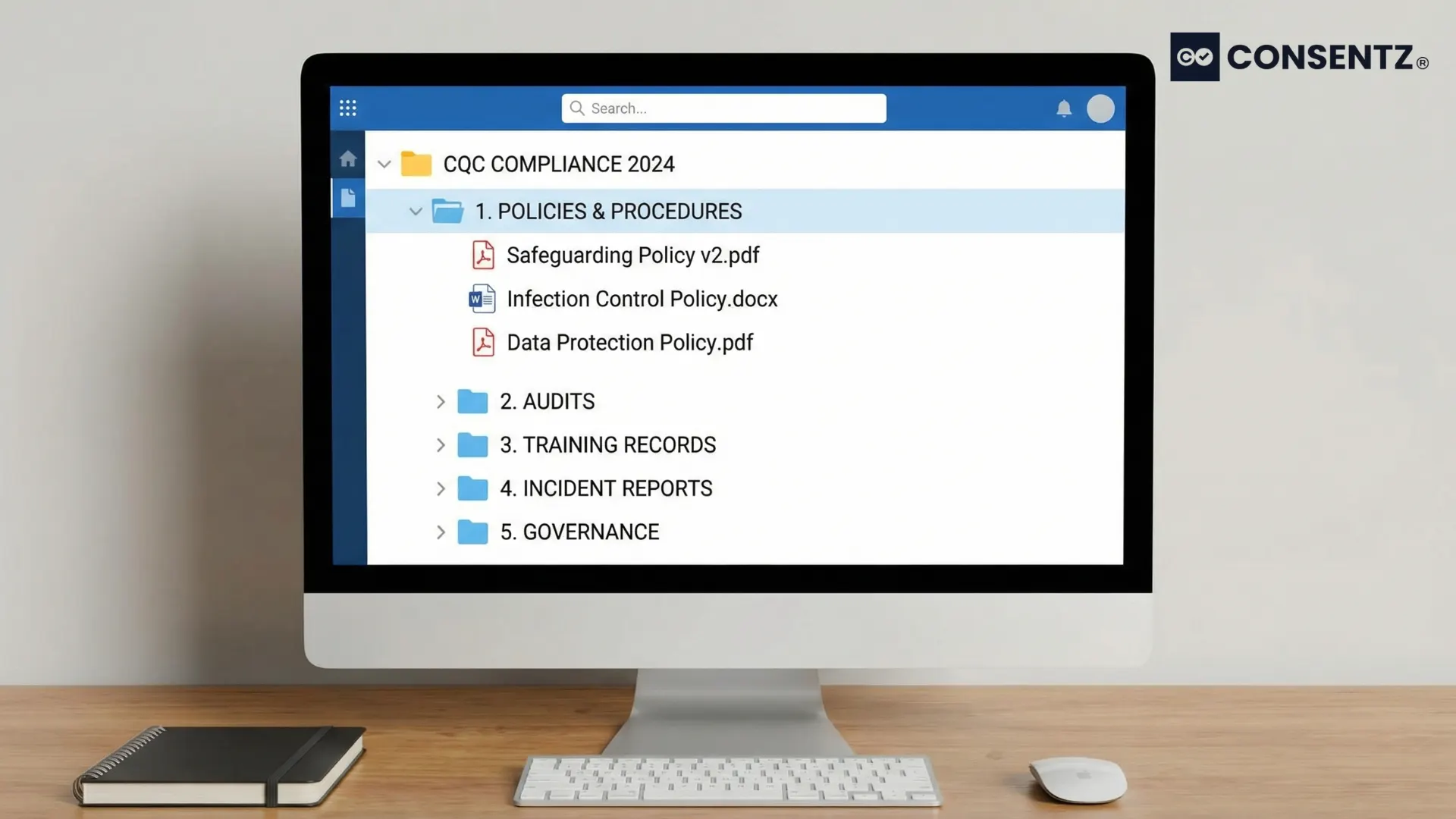
The Consentz CQC Compliance Module eliminates documentation chaos with 40+ professionally-written templates, automated reminders, and centralized storage—saving you hours of administrative work while ensuring inspection readiness.
Essential CQC Compliance Documents
Mandatory Policies
Safeguarding Policy – Protect vulnerable adults and children from abuse. Include local safeguarding board contacts, designated lead, reporting procedures, and staff training requirements. Review annually minimum.
Infection Prevention and Control Policy – Hand hygiene protocols, PPE usage, clinical waste disposal, equipment sterilization, and environmental cleaning schedules.
Consent Policy – Informed consent process including cooling-off periods (24-48 hours), capacity assessment, documentation requirements, and treatment-specific risk discussions.
Medicines Management Policy – Safe storage (temperature control, security), administration protocols, stock management, expiry monitoring, emergency medication procedures.
Complaints Handling Policy – How patients raise concerns, investigation procedures, response timelines, and learning from complaints. Must be visible to patients.
Duty of Candour Policy – Inform patients when things go wrong, apologize, investigate, and implement improvements. Legal requirement under Regulation 20.
Health and Safety Policy – Risk assessments, accident reporting, fire safety, first aid provision, staff welfare.
Required Risk Assessments
Clinical Risk Assessment – Risks for each procedure (vascular occlusion, infection, allergic reactions). Include risk ratings, mitigation strategies, and review dates.
Environmental Risk Assessment – Premises safety, including slip/trip hazards, fire risks, equipment hazards, accessibility, and security. Update when layout changes.
COSHH Risk Assessment – Hazardous substances (chemical peels, cleaning products, disinfectants). Detail safe storage, handling, disposal, and emergency procedures.
Consent Documentation
Treatment-Specific Consent Forms – Separate forms for each treatment covering specific risks, realistic expectations, alternatives, and costs. Essential for thread lifts, dermal fillers, Botox, chemical peels, lasers, and PDO threads.
Patient Information Sheets – Provide before consent appointments. Explain procedure details, recovery, risks, aftercare, and contact information.
Governance Documents
Statement of Purpose – Describes your service, including regulated activities, patient groups, staffing structure, and quality assurance. Update when services change.
Business Continuity Plan – Maintain services during disruptions (staff illness, equipment failure, premises issues).
Clinical Audit Schedule – Which audits you’ll conduct annually (infection control, consent, patient outcomes, record-keeping, medicines management).
Staff Documentation
Staff Training Records – Induction, mandatory training (safeguarding, infection control, emergency procedures), treatment-specific competency, CPD. Track expiry dates.
Staff Personnel Files – CV, qualifications, professional registration, DBS check, references, job description, appraisals, competency assessments.
Supervision and Appraisal Records – Regular supervision, performance reviews, professional development, and competency maintenance. Annual appraisals are a minimum.
Operational Records
Incident Report Forms – Document adverse events, near misses, accidents, and complaints. Include investigation findings, actions taken, and lessons learned.
Equipment Maintenance Logs – Servicing, calibration, and safety checks for all equipment. Critical for lasers, medicine fridges, autoclaves, and emergency equipment.
Stock Management Records – Temperature logs, batch tracking, expiry checks, stock reconciliation.
Patient Feedback Records – Document compliments, suggestions, and complaints. Show trend analysis and improvements implemented.
Managing all these documents manually is overwhelming. Consentz automates the entire process with customizable templates for every required document, automatic version control, and review reminders—ensuring nothing falls through the cracks.
Documentation Best Practices

Clinic-Specific Customization – Generic templates fail inspections. Add your clinic name, staff names, specific procedures, actual processes, and local contacts.
Keep Current – Review all policies annually minimum. Update when regulations change. Document review dates. Maintain version control.
Ensure Accessibility – Store documents in an organized central location. Ensure all staff know where to access them. Provide training on key policies.
Document Everything – If not documented, it didn’t happen. Record policy reviews, staff training, incident investigations, equipment maintenance, and clinical audits.
How Consentz Simplifies CQC Compliance Documentation
The Consentz CQC Compliance Module transforms CQC compliance documentation from overwhelming paperwork into an organized, automated system:
40+ Pre-Written Templates – Every essential document ready to customize: safeguarding policies, infection control procedures, treatment-specific consent forms, risk assessments, staff training records, incident reports, and more. Templates align with current CQC Fundamental Standards and include implementation guidance.
Centralized Cloud Storage – All compliance documents in one secure location accessible from any device. Organize by category, grant role-based access to staff, search documents instantly, and never lose critical files again.
Automated Version Control – Track every document change automatically. System maintains a complete history showing who made changes and when, flags outdated documents needing review, and ensures staff always access current versions—no more confusion about which policy is the latest.
Smart Review Reminders – Never miss deadlines again. Automated alerts 30 days before policy reviews are due, scheduled reminders for annual audits, equipment service due dates, and staff training renewal prompts. Set it once, forget it—the system handles the rest.
Inspection-Ready Reports in Seconds – Generate comprehensive documentation reports instantly showing all policies with review dates, staff training compliance rates, completed clinical audits, and document access logs. When inspectors arrive, demonstrate governance in minutes instead of scrambling through files.
Digital Signatures with Audit Trails – Staff and patients sign documents electronically with legally valid signatures. Full audit trail shows who signed, when, from which device, with IP verification. More secure than paper signatures and instantly accessible.
Treatment-Specific Consent Forms – Pre-built consent forms for thread lifts, dermal fillers, Botox, chemical peels, lasers, and more. Each form includes procedure-specific risks, cooling-off period documentation, capacity assessment, and realistic outcome expectations. Customizable to your clinic’s protocols.
Staff Training Management – Track mandatory training completion, record competency assessments, schedule refresher courses, and maintain CPD logs. Automated alerts when certificates near expiry ensure continuous compliance.
Multi-Location Support – Manage documentation across multiple clinic locations from one dashboard. Maintain location-specific details while sharing core policies. Perfect for growing practices.
Real Results: “Preparing for our first CQC inspection was terrifying—we had policies scattered everywhere, many outdated. Consentz organized everything in two weeks. When the inspector arrived, I pulled up every document she requested in seconds. She specifically praised our documentation systems. We achieved ‘Good’ on the first inspection.” — Dr. Sarah Mitchell, London Aesthetic Clinic
Time Savings: “I was spending 10-15 hours monthly managing compliance paperwork—updating policies, tracking staff training, remembering review deadlines. Consentz cut this to under 2 hours. The automated reminders mean nothing gets missed, and I can focus on patients instead of admin.” — James Patterson, Manchester Aesthetic Practice
Common Documentation Failures
Generic Templates – Using downloads without customization. Inspectors identify these immediately.
Outdated Documents – Policies not reviewed for 2+ years show poor governance.
Missing Treatment-Specific Forms – One generic consent form for all treatments. Each procedure needs specific documentation.
No Implementation Evidence – Beautiful policies staff have never seen. If staff can’t explain policies during interviews, documents are meaningless.
Incomplete Staff Records – Missing DBS checks, expired training certificates, and no competency assessments.
Frequently Asked Questions
1) How often should I review CQC compliance documents?
Review all policies annually, at a minimum. Clinical risk assessments need review after incidents or procedure changes. Staff training records require ongoing monitoring. Equipment logs need daily/monthly checks. Document every review with date and reviewer name.
2) Can I use the same policies for multiple clinic locations?
Core policies can be shared but must specify location-specific details like local safeguarding contacts, designated leads at each site, and premises-specific risk assessments. Each registered location needs its own Statement of Purpose.
3) What happens if I can’t produce a document during a CQC inspection?
Missing essential documents creates serious governance concerns. Inspectors may issue requirement notices, downgrade ratings, or suspend registration. Always have documents organized and immediately accessible.
4) Do I need paper copies, or are digital documents acceptable?
Digital documents are acceptable. Ensure backups exist, staff can access systems easily, and you can print if needed. Some inspectors may request printed copies during site visits.
5) How long should I keep CQC compliance documents?
Maintain policies for the life of registration plus 3 years. Keep patient records 8 years minimum (longer for children—until 25th birthday). Staff records retain 6 years after employment ends. Equipment logs are kept for the equipment’s lifetime plus 3 years.
Conclusion
Comprehensive CQC compliance documentation proves you meet regulatory standards. Organized systems save time during inspections, reduce compliance stress, and demonstrate professional governance.
Transform your CQC compliance documentation from chaos to confidence. The Consentz CQC Compliance Module provides everything you need:
✅ 40+ professionally-written templates covering every required document
✅ Automated reminders ensuring you never miss review deadlines
✅ Centralized cloud storage accessible 24/7 from any device
✅ Inspection-ready reports generated in seconds
✅ Digital signatures with complete audit trails
✅ Multi-location support for growing practices
✅ Free implementation support and training
Join 500+ UK aesthetic clinics using Consentz to streamline CQC compliance. Stop struggling with scattered documents, missed deadlines, and inspection anxiety. Get organized, stay compliant, and focus on what matters—delivering exceptional patient care.
References
Care Quality Commission. (n.d.). Guidance for providers. Retrieved from https://www.cqc.org.uk/guidance-providers
Care Quality Commission. (n.d.). Regulations for service providers and managers. Retrieved from https://www.cqc.org.uk/guidance-providers/regulations-enforcement/regulations-service-providers-managers